Retatrutide tracker
for iPhone.
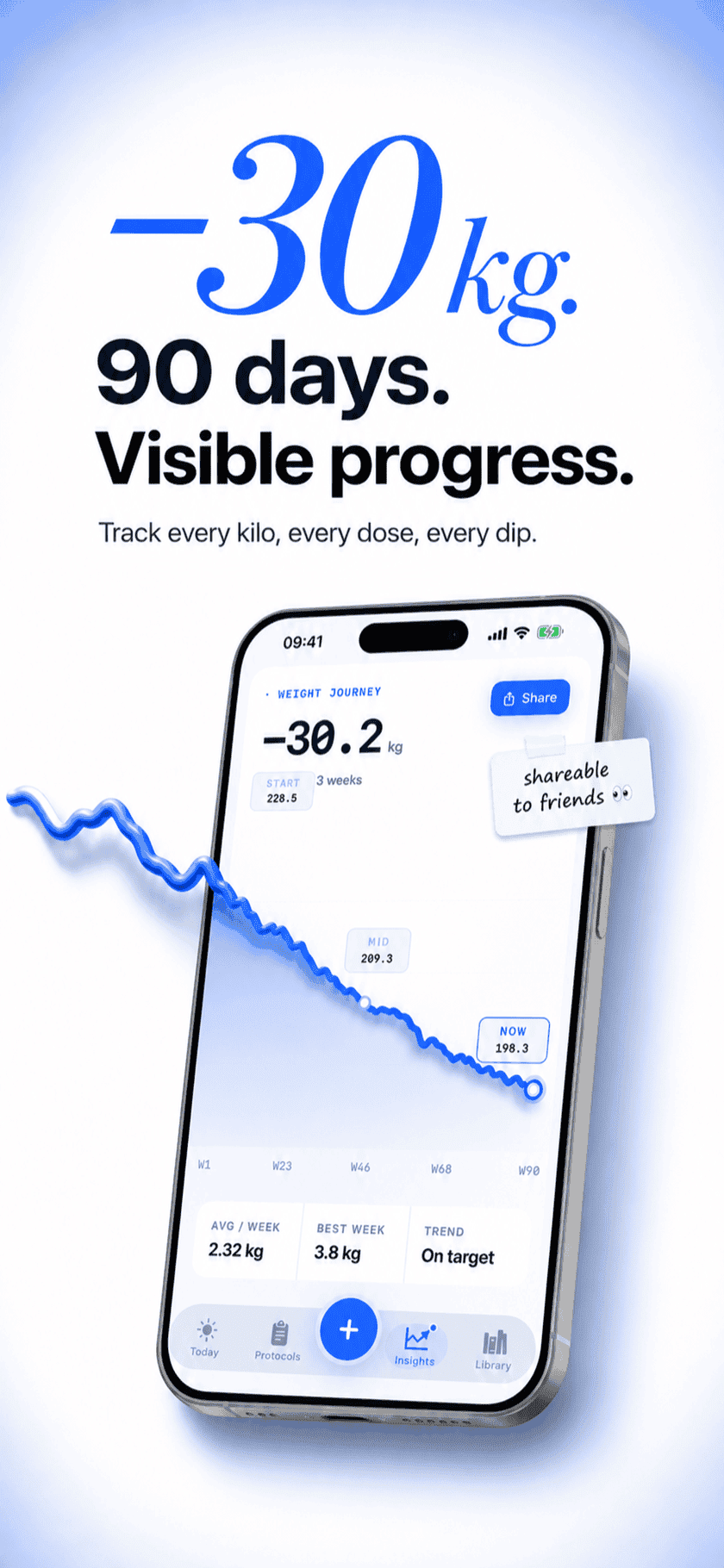
Retatrutide is the newest of the major GLP-1 family members and the first triple agonist to make trial waves. The cadence is familiar — once a week, escalate every four — but the molecule is potent and the dose ladders run higher than the older drugs. Peptide Protocol gives Retatrutide its own preset, holds the schedule, and overlays your weight chart against the dose timeline so the bigger jumps don't get away from you.
Download Peptide Protocol — FreeRetatrutide at a glance
Quick reference for the protocol-builder. Retatrutide is investigational — confirm any protocol design with a licensed clinician.
| Class | GLP-1 / GIP / glucagon triple receptor agonist |
|---|---|
| Brand names | None — investigational compound (Eli Lilly, Phase III as of 2026) |
| Plasma half-life | ~6 days |
| Typical cadence | Once weekly, subcutaneous |
| Trial dose ladder | 2 mg → 4 mg → 8 mg → 12 mg, escalating every 4 weeks (Phase II / III protocol pattern) |
| Common reconstitution | 5 mg vial + 1.0 mL bacteriostatic water → 5 mg/mL → 0.40 mL = 2 mg dose |
| Insulin syringe | U-100 most common (40 units = 0.40 mL = 2 mg at 5 mg/mL) |
| Stack pairings | Generally run alone — its glucagon-axis activity already covers more of the metabolic surface than mono-GLP-1 |
| Common side-effect pattern | Nausea, GI discomfort, sometimes elevated heart rate at higher doses; trials report stronger appetite suppression than dual-agonist comparators |
For trial-phase results, side-effect literature, and a full comparison against Tirzepatide and Semaglutide, see the Retatrutide guide on peptide-calc.app.
Built around Retatrutide protocols
Three things the app does that matter especially for a fast-escalating, high-potency molecule.
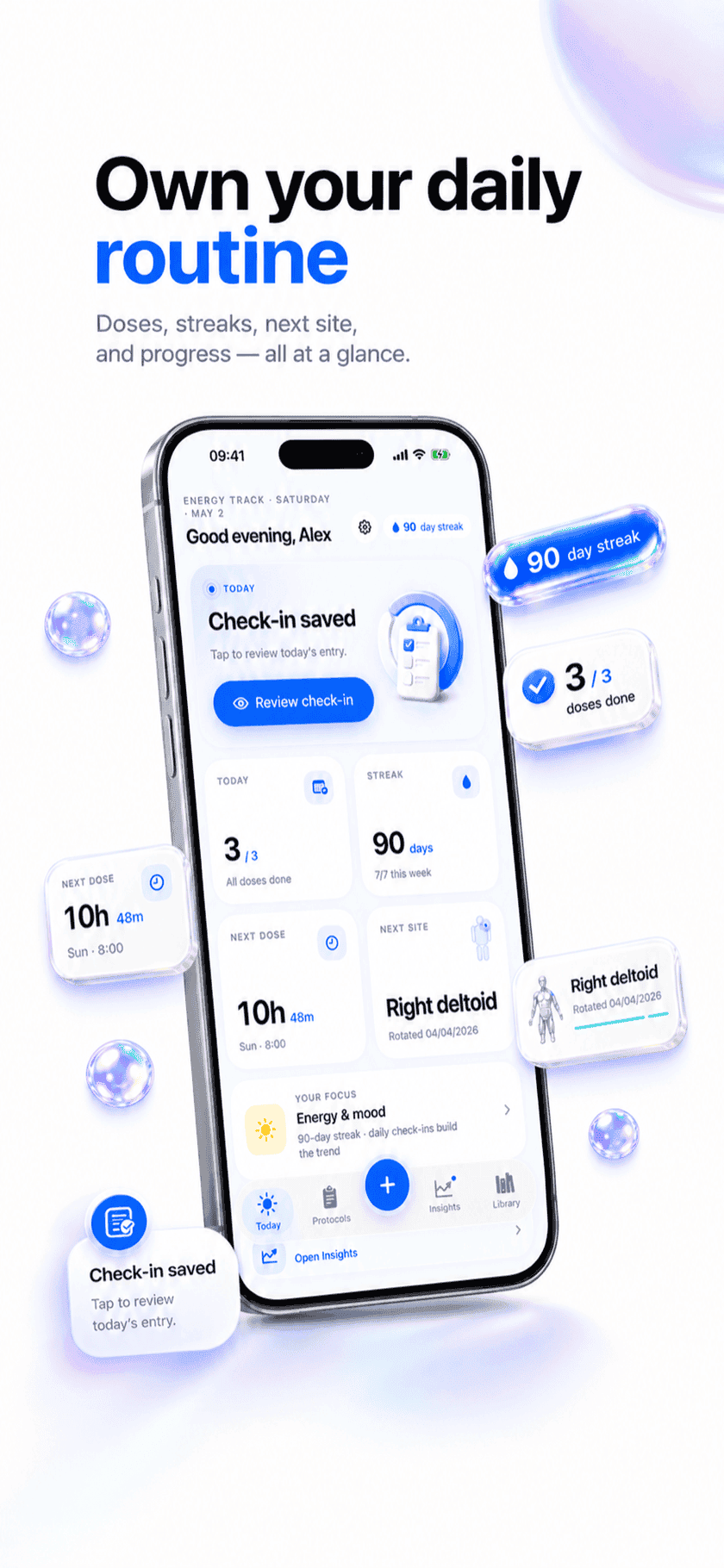
Weekly cadence with custom ladder
No FDA label means no fixed ladder. Build your own: 2 → 4 → 8 → 12 mg with whatever hold length you decided with your clinician. The app advances on schedule and updates the calculator the same moment.
Math for the bigger doses
12 mg per week is a lot of solution to draw. The calculator handles 5 mg/mL and 10 mg/mL concentrations and shows the unit count on every common syringe — useful when the volume gets close to the syringe's max.
Heart rate + GI in the check-in
Triple agonism brings a different side-effect profile. The daily check-in includes free-text notes; the weekly report aggregates them next to the dose log so a slow-rising HR or appetite shift doesn't go unnoticed.
What it looks like

Retatrutide tracking — common questions
Does Peptide Protocol support Retatrutide?
Yes. Retatrutide is a first-class preset in the calculator. Pick the vial mass and the BAC water volume you used, set your target dose, and the app outputs the unit count for a U-100, U-50, or U-30 insulin syringe — same engine used for Tirzepatide and Semaglutide protocols.
What escalation pattern do I use for Retatrutide?
Retatrutide is still in clinical trials, so there is no FDA-labeled escalation. Many self-protocols mirror the trial design: 2 mg → 4 mg → 8 mg → 12 mg, holding each step for 4 weeks. The app supports any custom ladder you define — confirm dosing with a licensed clinician before you build it.
How is Retatrutide tracking different from Semaglutide or Tirzepatide?
Mechanistically Retatrutide adds a glucagon-receptor agonist on top of GLP-1 and GIP, which often shows up as stronger thermogenesis and a different side-effect curve. The app handles all three the same way — weekly cadence, escalation steps, weight overlay, GI symptom log — so you can switch protocols without re-learning the workflow.
Related peptide trackers
Track your Retatrutide on your iPhone.
Free on the App Store. Builds your first protocol in under five minutes. No account required.
Download on the App StoreNot medical advice. Retatrutide is an investigational compound. It has not been approved by the FDA or other major regulators for any indication and is not available by prescription as of 2026. The information on this page is informational, drawn from peer-reviewed Phase II / III trial literature, and is not a recommendation to use the compound. Always consult a licensed healthcare professional before starting, modifying, or stopping any compound — see the full Retatrutide reference for primary sources.